Engineered Protein Binds to Alpha-synuclein to Prevent Toxic Clumping, Study Reports
Written by |
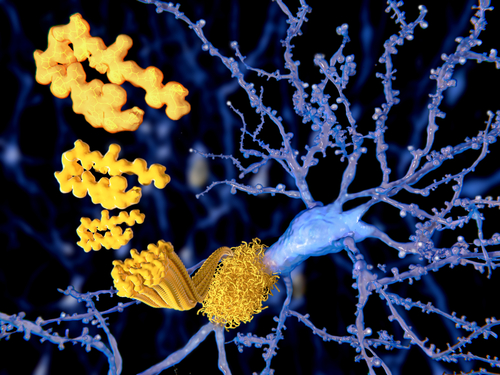
An engineered protein known as AS69 is able to bind to individual units of alpha-synuclein to prevent them from clumping, and — in a fly model of Parkinson’s disease — its use led to preserved motor function, a study reports.
The study, “An engineered monomer binding-protein for α-synuclein efficiently inhibits the proliferation of amyloid fibrils,” appeared in the journal eLIFE.
Cellular aggregates, or clumps, of the protein alpha-synuclein are an established hallmark of Parkinson’s and focus of research into treatments. “The link between [alpha]-synuclein aggregation and PD [Parkinson’s] has been known for two decades … however, translation of this scientific discovery into a therapy has proven challenging,” the scientists wrote.
A team of international researchers now engineered a binding protein, known as beta-wrapin AS69, that can bind with high affinity to monomers (single units) of alpha-synuclein and induce a specific conformational or structural change called a beta hairpin.
The alpha-synuclein region that adopts this structure is essential for its clumping, as indicated by the presence of a cluster of disease-related mutation sites. Upon binding to this region, AS69 stops alpha-synuclein from aggregating into amyloid fibrils.
To better determine the potential of AS69 as a therapy, the scientists also tested it in cellular and animal models.
In vitro, the team confirmed that AS69 specifically worked to lower alpha-synuclein aggregation and not its amount, testing both with wild-type (normal) protein and a variant (A53T) previously linked to familial Parkinson’s and to quicker protein clumping. (In vitro refers to experiments in lab dishes; in vivo experiments are those within a living organism, including animal models.)
In fruit flies with A53T alpha-synuclein in their brain nerve cells, AS69 was then seen to preserve the flies’ ability to climb (reflecting motor function), which was associated with fewer alpha-synuclein aggregates. This climbing ability progressively declined in the absence of AS69.
Protein aggregation is a complex process involving multiple microscopic steps. It starts with an event called primary nucleation, in which misfolded (altered shape) proteins clump together to form fibrils, which then elongate. This first step proceeds slowly, potentially taking up to several decades.
A later event is called secondary nucleation. Here, aggregation speeds up and exponential growth occurs, with existing clumps promoting the formation of new ones. This faster phase is associated with evident disease, and a potential for rapid progression.
When the team investigated specific steps of alpha-synuclein protein clumping, it found that fibril elongation was suppressed by AS69 in a concentration-dependent manner. Both fibrils and AS69 competed for the single units of alpha-synuclein, but while the interaction with AS69 occurred within seconds, binding to fibrils took minutes to hours.
In contrast, the interaction of free AS69 with fibrils was weak, if it existed at all.
AS69 was also found to interfere with lipid (fat)-induce alpha-synuclein aggregation, and was a more efficient inhibitor of the amplification of alpha-synuclein amyloid fibrils than beta-synuclein — a protein known to suppress alpha-synuclein clumping. Importantly, by binding to alpha-synuclein, AS69 also prevented secondary nucleation.
Based on these results, the team proposed that the complex of AS69 with alpha-synuclein incorporates into a fibril precursor, and prevents this precursor from undergoing the structural changes needed for further aggregation.
“An inhibitor functioning according to this dual mode, that is being active both as a free molecule and as a complex with (…) [alpha]-synuclein, is expected to efficiently reduce [alpha]-synuclein aggregation in vivo,” the researchers concluded.

Jb
Anyone reading this today will most likely never receive any benefits of this (if it ever does proceed) still on .... fruit flies ......worms ...... mice.
Last real discovery to help those with Parkinson’s was in the 1960s - levodopa Nothing if real significance developed since I guess the question that follows- why no results ? I was once told “researchers” are really more about being published than making discoveries. Levodopa isn’t a cure and for some isn’t at all effective
BENNY BURKE
Big Pharma----- squash