Scientists Use New Tool to Study How Nerve Cells Interact to Control Movement
Written by |

Using a new tool, researchers were able to see how two different sets of neurons interact in mice to control movement. They believe the method, called spectrally resolved fiber photometry, may help in unraveling what goes wrong in the brains of Parkinson’s patients and those with other disorders.
The research, “Spectrally Resolved Fiber Photometry for Multi-component Analysis of Brain Circuits,” was published in the journal Neuron.
Progressive damage to nerve cells in the substantia nigra region of the brain lowers levels of the neurotransmitter dopamine — a chemical responsible for communication between neurons, or nerve cells — and is considered a hallmark of Parkinson’s disease.
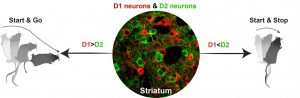
Clinical studies in Parkinson’s patients and preclinical research in monkeys suggests that loss of dopamine causes an imbalance in the activity of two groups of neurons: the direct pathway (D1) and indirect pathway (D2). However, this hypothesis could not be confirmed experimentally due an inability to accurately distinguish between these cell types in the brain.
Using spectrally resolved fiber photometry (SRFP), a tool developed at the National Institutes of Health (NIH), researchers in an NIH office labeled D1 and D2 neurons with green and red fluorescent sensors and were then able to effectively follow how they work together in neurons of living mice.
“Our method allowed us to simultaneously measure neural activity of both pathways in a mouse as the animal performed tasks,” Guohong Cui, MD, PhD, the study’s senior author, said in a press release. “In the future, we could potentially use SRFP to measure the activity of several cell populations utilizing various colors and sensors.”
The scientists observed that when activity in D1 was stronger than in D2 neurons, the animals did a “start and go” — starting movement and moving to another location. When D2 neuronal activity was stronger, a mouse does a “start and stop” — it initiates a movement, but stops soon after.
Both movements are normal in mice and their analysis may help predict what type of movement will be made based on the neural activity seen. Importantly, being able to trace such activity may help in understanding movement in mouse models of Parkinson’s.
“Based on these observations, we hypothesize that the direct-pathway (D1) activation serves as a movement start signal, and its magnitude determines the vigor of a movement. Meanwhile, the concurrently activated indirect pathway (D2) serves as a scalable stop signal that determines whether the initiated movement will continue or be terminated,” the researchers wrote.
Unlike current methods that cannot distinguish which neurons are generating an electrical output, “SRFP is more specific, because we can distinguish between groups of neurons and see their activity,” said Chengbo Meng, PhD, one of the study’s lead authors.
“We have developed a novel … method for simultaneous multi-color fluorescent signal measurement and unmixing from deep brain structures in vivo,” the study states. “Using this method, we show for the first time that the neural activities of two parallel … pathways are highly synchronized, and the magnitude of activation in these two pathways collaboratively determines the dynamics and fate of movement.”
In addition Parkinson’s disease, the team believes SFRP will contribute to a better understanding of Alzheimer’s, multiple sclerosis, stroke and addiction.



rameshkaul
whether personal genome test/dna- ngf test or similar dna tests will be useful for early detection of PD symptoms?