Understanding Metabolic Changes in Parkinson’s Disease Could Lead to New Treatments
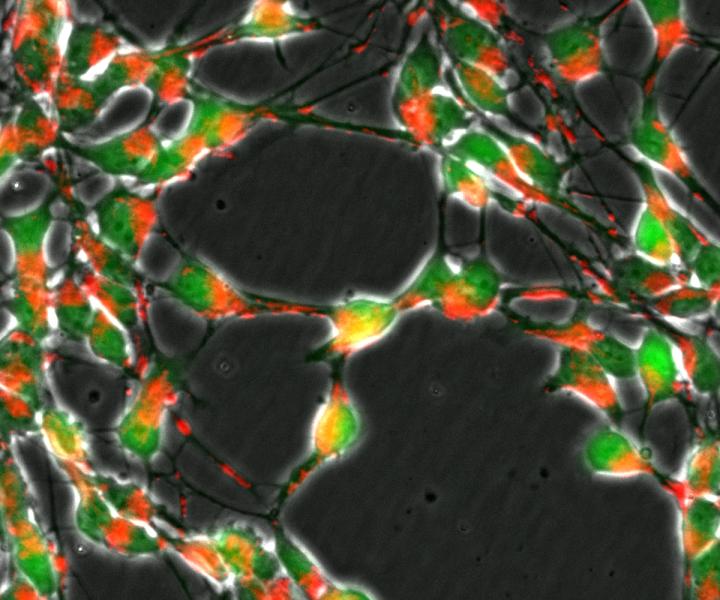
Loss of the DJ1 gene in nerve cells (green) impairs the transport of mitochondria (red) and important metabolic pathways of the cellular respiration. CREDIT University of Luxembourg
Novel research could advance the basic understanding of the genetic causes behind Parkinson’s disease (PD). Researchers have examined the defective DJ1 (PARK7) gene, which controls cellular metabolism and may play a role in Parkinson’s, and their report, “Loss of DJ-1 impairs antioxidant response by altered glutamine and serine metabolism,“ appeared in the journal Neurobiology of Disease.
Parkinson’s disease is caused by a loss of dopamine-producing cells in the substantia nigra, a brain region that controls the start and stopping of movement. People with Parkinson’s develop problems with movement such as freezing, gait problems, tremors, and rigidity. Dopamine cell loss might be caused by a process known as oxidative stress during which cells produce harmful free radicals faster than the body can remove them. This can cause cell death in the nervous system.
Researchers have found that mutations in the DJ-1 gene, coding for a molecule that prevents oxidative stress, can increase the risk of developing Parkinson’s. The current study sought to further understand how DJ-1 can affect cells and possibly play a role in the development of this disease.
The research team, led by Dr. Johannes Meiser of Luxembourg Centre for Systems Biomedicine at the University of Luxembourg, grew mouse neurons in vitro (in the lab) that specifically lacked the DJ gene. They found that the cells could not make crucial amino acids that are necessary to prevent oxidative stress. The neurons did not take up the glutamine that is needed for the synthesis of both the neurotransmitter glutamate, as well as the amino acid serine. Disruptions in this pathway affected antioxidant production as well.
“Without DJ1, neurons cannot absorb enough glutamine and this affects serine production,” Meiser said in a news release. “Both amino acids are important for producing glutathione, which is used to neutralize free radicals. In the absence of DJ1, this defense mechanism does not work effectively and oxidative stress occurs. This prematurely ages the cells.”
According to the scientists, the microglia — the immune system cells of the brain — seemed to contribute to this oxidative stress. Taken together, all of these effects could explain how Parkinson’s-induced cell death occurs.
The investigators plan to further study how these findings might be used to develop treatments for Parkinson’s disease. Specifically, if the observed changes in glutamine and serine can be altered, it could provide a target for Parkinson’s medications. Also, targeting oxidative stress could be another avenue for potential therapies.
Image credit: University of Luxembourg






