Structure of Toxic Protein Aggregates Opens Door to Novel Parkinson’s Disease Treatments
Written by |
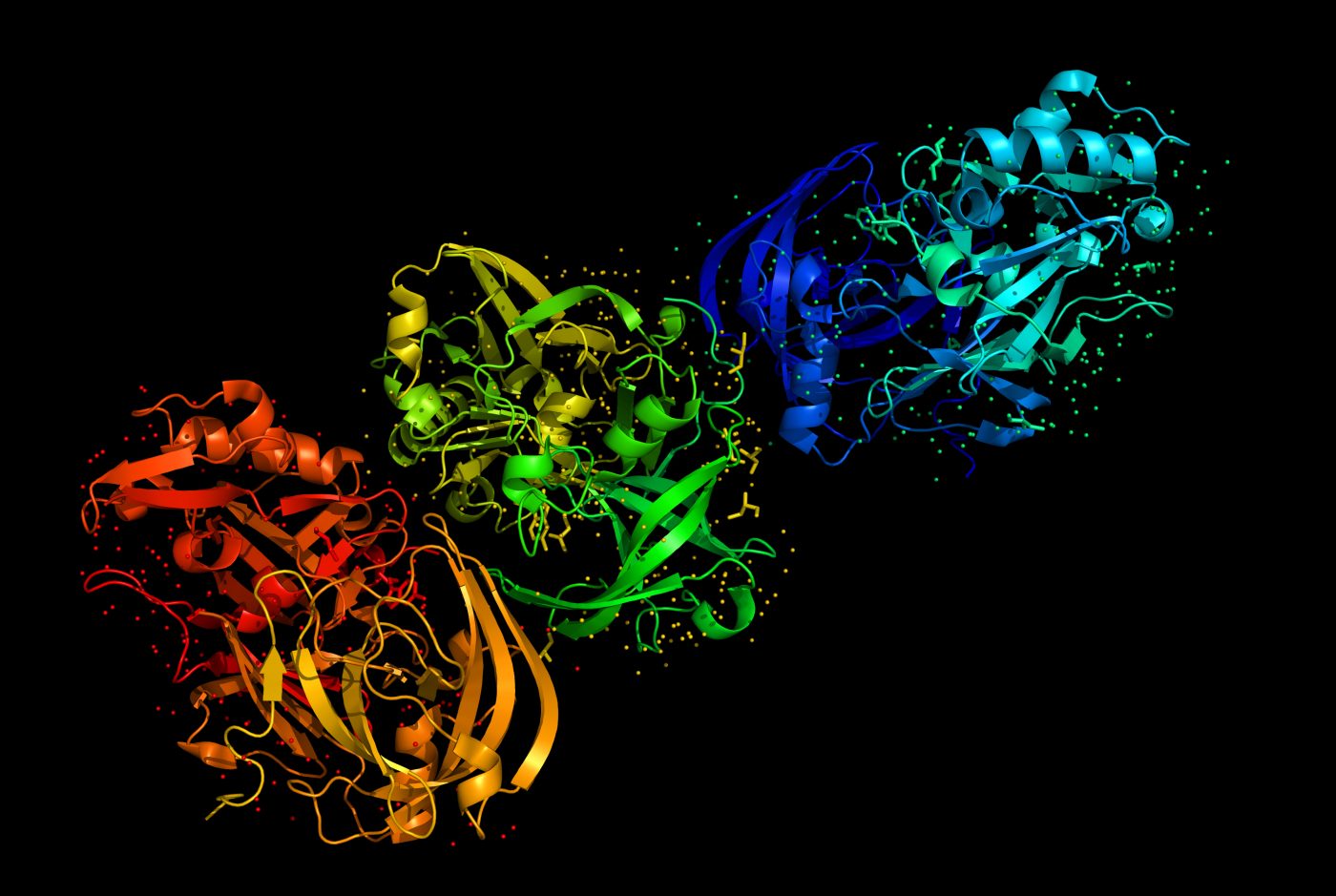
In a recent study led by researchers from the UCLA-DOE Institute entitled “Structure of the toxic core of α-synuclein from invisible crystals” published in Nature, an ultra-high-resolution structure of crystals from the core of alpha-synuclein fibrils, toxic assemblies formed in Parkinson’s disease patients, is presented. These insights may help in the development of effective drugs against this pathology.
Parkinson’s disease is neurodegenerative disorder that mainly affects neurons that control body movements. Central to its development is the accumulation of insoluble protein aggregates called Lewis bodies, which are mainly composed by alpha-synuclein. These assemblies are present in about 95% of patients with Parkinson’s, and are associated with neuronal death.
Alpha-synuclein is a 140 aminoacids protein abundant in neurons, mainly at their nerve terminals where it seems to contribute for synaptic signaling. Under pathological conditions, alpha-synuclein aggregates with its soluble counterparts, forming toxic insoluble oligomers and fibrils, which can reach several microns in length and 5-10 nanometers in diameter. Similarly with neurodegenerative amyloid aggregates, alpha-synuclein fibrils are characterized by adopting a linked beta-sheet configuration, whose structures were yet not fully known.
Using a novel method based on cryo-electron microscopy termed MicroED, the research team was able to obtain ultra-high-resolution structures (1.4 ångström resolution) of alpha-synuclein segments though to form the core and nucleation site of protein fibrils from a variant with a Parkinson’s disease-causing mutation.
Structures showed that fibrils are organized as pairs of beta-sheets, each having parallel beta-strands and antiparallel strands between the sheets. The zipper region between paired sheets is longer than in similar structures and each pair of beta-sheets contains two water molecules, whereas usually these are dry.
These findings suggest that this region might be a promising target of Parkinson’s disease therapies, encouraging the development of molecules that act on this zipper structure to inhibit alpha-synuclein fibrils formation. A similar approach has been used to successfully hinder the assembly of toxic protein aggregates associated with Alzheimer’s disease. However, according to structural biologist Yifan Cheng, who was not involved in the study, “it remains to be seen whether this model can account for the effects of other mutations in this region that cause Parkinson’s disease”.

Leave a comment
Fill in the required fields to post. Your email address will not be published.